
The Importance of Trust, Diversity and Equity in Clinical Trials
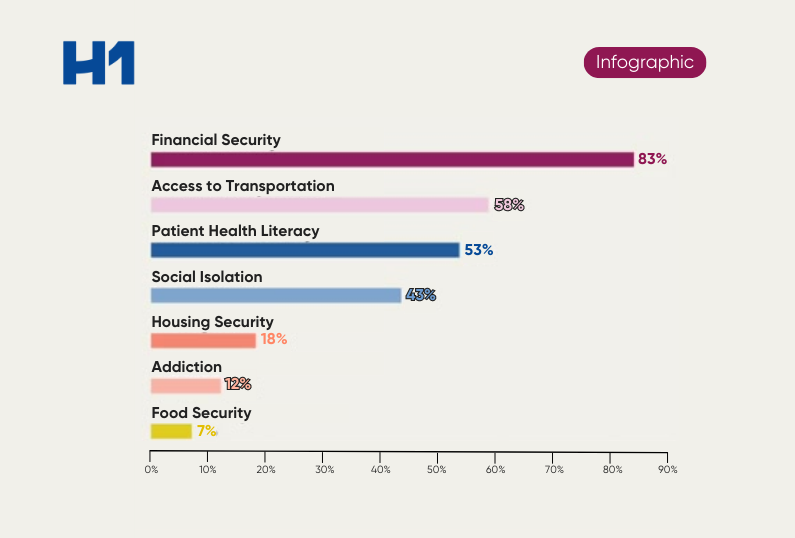
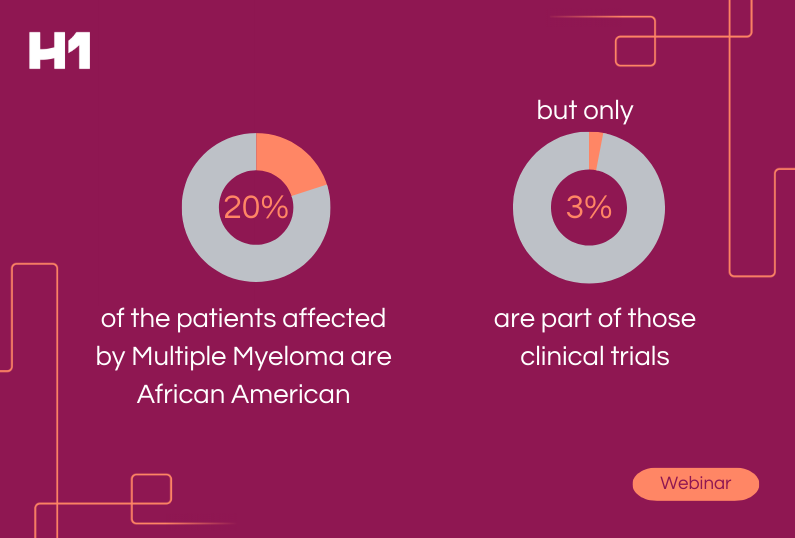
Clinical trials test the safety and efficacy of treatment options in drug therapies, medical devices, and therapeutics for patients and establish new standards of care. Much like the healthcare industry, biopharmaceutical and life sciences companies have lacked in efforts to incorporate diversity and inclusion in trial investigators, sites, and participants.
Download this resource to learn how data intelligence and analytical insights based on clinical expertise and diversity focus are changing the way companies optimize drug development from trial success to commercial launch.
Get your copy of the resource guide today.















































































 HCP Universe
HCP Universe Trial Landscape
Trial Landscape